Protease Inhibitor Cocktail
Features
- Wide range - contains inhibitors for a variety of serin and cysteine protease
- Contain no EDTA - efficient for purification of metalloproteins and His tag gene exchanged proteins
- Easy to use - dilute according to your need (100x stock concentration)
Product Information
Inhibition of intra and extra cellular proteases is vital to purify and collect the expressed proteins. Saving trouble of finding adequate inhibitors, a wide range of proteases are inhibited by the Protease Inhibitor Cocktail. This product does not interfere with metalloproteins since it is EDTA free.
Composition
| Inhibitors | #04080-11 for General Use |
#03969-21 EDTA free |
#25955-11 for Mammalian |
|---|---|---|---|
| 4-(2-Aminoethyl) benzenesulfonyl fluoride hydrochloride (AEBSF) | √ | √ | √ |
| Aprotinin | √ | √ | √ |
| E-64 | √ | √ | √ |
| Leupeptin hemisulfate monohydrate | √ | √ | √ |
| Disodium dihydrogen ethylenediaminetetraacetate dihydrate |
√ | ||
| Bestatin | √ | ||
| Pepstatin A | √ |
Applications
Comparison of EDTA containing and EDTA free inhibitor activity for Thermolysin
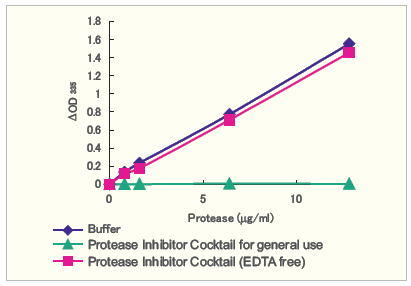
Figure 1. Thermolysin (from Bacillus thermoproteolyticus) is a metalloprotease. The compared protease inhibitors do not inhibit Thermolysin activity. The presence EDTA in the common product inhibits Thermolysin, while the EDTA free product does not cause damage.
| Thermolysin (mg/ml) | ||||
|---|---|---|---|---|
| 0.8 | 1.6 | 6.4 | 12.8 | |
| Protease Inhibitr Cocktail for General Use | 100.0 | 99.6 | 99.2 | 99.7 |
| Protease Inhibitr Cocktail (EDTA free) | 9.7 | 8.2 | 8.0 | 6.3 |
Figure 2. The decrease in Thermolysin activity (in %) in the presence of protease inhibitor cocktails.
Protocols
Protease Inhibitor Cocktail (EDTA free)![]() (PDF 86 KB)
(PDF 86 KB)
References
- T Sunabori et al.: The American Journal of Pathology(2016)
- K Hiramatsu et al.: British journal of cancer (2016)
- S Kawauchi et al.: Journal of pharmacological sciences 124.2 (2014): 180-191
- Y Ariumi et al.: Journal of virology 85.14 (2011): 6882-6892
- Okada, S. et al. The Journal of Cellular Physiology 226(2), 552-558 (2011)
- Yang, JH. et al. The Journal of Biological Chemistry (2010)
- Iyama, T. et al. Nucl. Acids Res. 38(14), 4834-4843 (2010)
- Kimura, Y. et al. Cancer Research 70(2), 501-511 (2010)
- H Kimura et al.: Cell structure and function 33.1 (2008): 61-73
- Y Naito et al.: Molecular and cellular biology 27.8 (2007): 3008-3022
- Y Tokuzawa et al.: Molecular and cellular biology 23.8 (2003): 2699-2708
- Burnett, T. J. et al. J. Bacteriol 165, 139-145 (1986)
- Hagiwara B et al. J. Biochem., 45, 185-194 (1958)
Downloads
Phosphatase Inhibitor Cocktail (EDTA free) / Protease Inhibitor Cocktail (EDTA free)![]() (PDF 557 KB)
(PDF 557 KB)
Ordering information
(Storage) RT: Room temperature, A: Cool and dark, R: Refrigerator, F: Freeze