ACROBiosystems社 COVID-19 R&D: A YEAR IN REVIEW
 |
||
 |
||
The Process of How SARS-CoV-2 Infects the Host
|
||
|
||
 |
Positive Phase 1/2 Study Results of Pfizer COVID-19 mRNA Vaccine FDA approves the Pfizer vaccine for emergency use in the US on December 11. It was a significant milestone to get authorization of a vaccine within a year of scientists identifying the new coronavirus. Let’s review their phase 1/2 study again. Read More >>> |
|
|
|
|
|
|
||
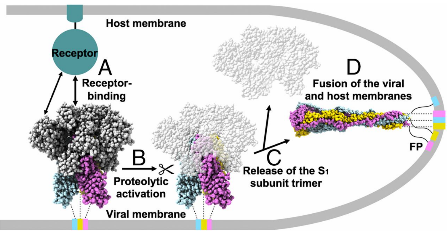 |
Recombinant Spike Protein Trimer: more powerful than you think
S protein is considered the key to the infection process. Let’s see how the spike trimer protein could help in COVID-19 diagnostics, vaccines, and antibody development. Read More >>> |
|
COVID-19 anti-nucleocapsid antibodies verified with inactivated virus samples |
||
 |
For the development of the antigen test, scientists are currently facing a dilemma regarding the best method of selection for an appropriate sample for the assay development. It is clear that the better sample is the patient's nasopharyngeal swab but due to biosafety reasons, it can be difficult to obtain access to patient samples early in the discovery stage. It could be a better way to assess an antigen test is conducted by utilizing an inactive virus for the assay development. Read More >>> |
|
ACROBiosystems and the Fight Against COVID-19 In this interview, Dr. Yinan Jiang discusses ACROBiosystems' efforts in the fight against COVID-19 and the search for a vaccine.
>>> Read how ACROBiosystems has been working on the fight against COVID-19 |
||
Fount of Information は、新商品、新規取扱メーカーなどの情報をいち早く紹介するコンテンツです。情報発信のスピードを重視しているコンテンツのため、現時点で法規制や取り扱いを確認できていない商品、定価を設定できていない商品があります。ご要望やご照会を受けた商品について、法令整備や在庫の充実を図ります。