ACROBiosystems社 ASCO 2022 - Daiichi's third conjugate progresses in breast and lung cancer
|
この製品に関するご意見・ご照会・お問合せはこちら
| Insight on ADCs at ASCO 2022 |
| ASCO 2022 – Daiichi's third conjugate progresses in breast and lung cancer
Since Enhertu's successes, the next antibody-drug conjugates (ADC),Patritumab deruxtecan, emerging from Daiichi Sankyo's pipeline, has been closely watched. Patritumab deruxtecan (HER3-DXd) includes a human anti-HER3 monoclonal antibody covalently bound to a topoisomerase I inhibitor payload via a stable tetrapeptide-based cleavable linker, which interrupts DNA replication in cancer cells.
Patritumab deruxtecan (HER3-DXd) is active in patients with heavily pretreated HER3-expressing metastatic breast cancer, according to results of a phase 1/2 study presented during the 2022 ASCO Annual Meeting by Ian E. Krop, MD, PhD, with a confirmed objective response rate (ORR) of 30% and a toxicity-related discontinuation rate of 10%. Besides, HER3-DXd have Efficacy in Lung Cancer. In December 2021, the FDA granted a breakthrough therapy designation to Patritumab deruxtecan for the treatment of patients with metastatic or locally advanced EGFR-mutated non–small cell lung cancer (NSCLC) with disease progression on or after treatment with a third-generation TKI and platinum-based therapies.
|
To meet the needs of ADCs development, ACROBiosystems can provide:
★ A variety of high-quality target proteins;
★ SPR/BLI analytical service;
★ MMPs/Cathepsin/uPA for cleavable linker;
★ Anti-payload antibodies, anti-idiotypic antibodies(ADA) for immunogenicity and PK analysis.
Learn more about related products about ADCs development
Verified protocol offered for free!
 Download the eBook: Key point of ADCs design Download the eBook: Key point of ADCs design
|
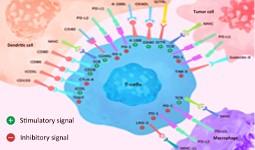 Immune Checkpoint Proteins Immune Checkpoint Proteins

|
 GMP Grade Cytokines GMP Grade Cytokines

|
 Multi-pass Tps Multi-pass Tps

|
|
Fount of Information は、新商品、新規取扱メーカーなどの情報をいち早く紹介するコンテンツです。情報発信のスピードを重視しているコンテンツのため、現時点で法規制や取り扱いを確認できていない商品、定価を設定できていない商品があります。ご要望やご照会を受けた商品について、法令整備や在庫の充実を図ります。