ACROBiosystems社 Scalable Manufacturing of CAR cells
|
この製品に関するご意見・ご照会・お問合せはこちら
 
|
|
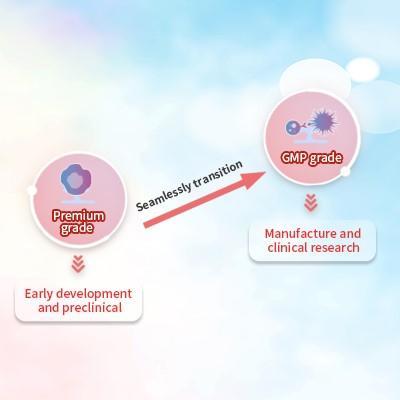
Scalable Manufacturing Platforms for CAR T
As the demand for CAR T cell therapy increases, scalable manufacturing platforms will be a critical component in preventing manufacturing bottlenecks. By embracing the principle of ‘quality by design’, our raw materials are designed to help manufacturers achieve robust and reproducible CAR-T cell production based on manufacturing science and quality risk management.
|
|
|
|
|
|
GMP grade GENIUS™ Nuclease
Using GMP-grade nucleases ensures that your final therapeutic does NOT have any residual DNA/RNA while following current manufacturing guidelines.
|
 |
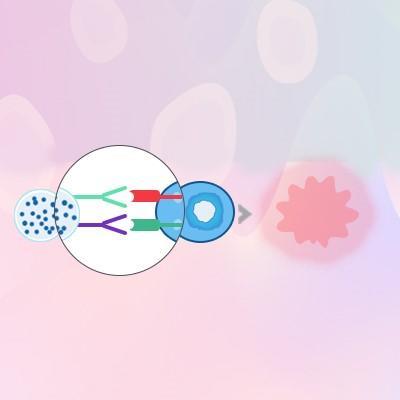 |
CD3/CD28 Activation Beads
An Easier, Smarter Way to Stimulate and Expand Your T Cells
- Diverse cell media compatibility with common media.
- Improved naïve and stem memory T, cell count.
- No required preparation steps like feeder cells, APCs, or antigen preparations.
|
|
GMP Grade Anti-CD3 Antibody(OKT3)
OKT3 antibodies are critical for the activation and expansion of CD3+ T cells in research and ex vivo cell culture applications. Our GMP-grade OKT3 antibodies are designed to be for use under CMC and clinical research.
|
 |
 |
CRISPR-Cas Nuclease
Our Genpower series of nucleases now includes Cas9 and Cas12a. These proteins are mainly used for CRISPR-Cas system-targeted gene editing with high editing efficiency.
|
|
Fount of Information は、新商品、新規取扱メーカーなどの情報をいち早く紹介するコンテンツです。情報発信のスピードを重視しているコンテンツのため、現時点で法規制や取り扱いを確認できていない商品、定価を設定できていない商品があります。ご要望やご照会を受けた商品について、法令整備や在庫の充実を図ります。