ACROBiosystems社 Treatment of Cancer Improved by Combination Therapy Regimen
 |
|||||||||||||
|
|||||||||||||
|
Treatment of Cancer Improved by Combination Therapy Regimen
The Phase 3 trial of Sanofi and Regeneron’s PD-1 inhibitor Libtayo in combination with platinum-doublet chemotherapy was stopped early after meeting its overall survival (OS) primary endpoint in patients with advanced non-small-cell lung cancer (NSCLC). Besides, A new preclinical study suggests that combining immune checkpoint inhibitors targeting PD-1/ PD-L1 with MAPK-targeted therapy—drugs typically used separately— may help overcome cancer treatment resistance.
More and more studies have shown that immune checkpoint combination therapy has a good effect in dealing with cancer.
ACROBiosystems has developed an exclusive immune checkpoint protein collection.
Product Features: 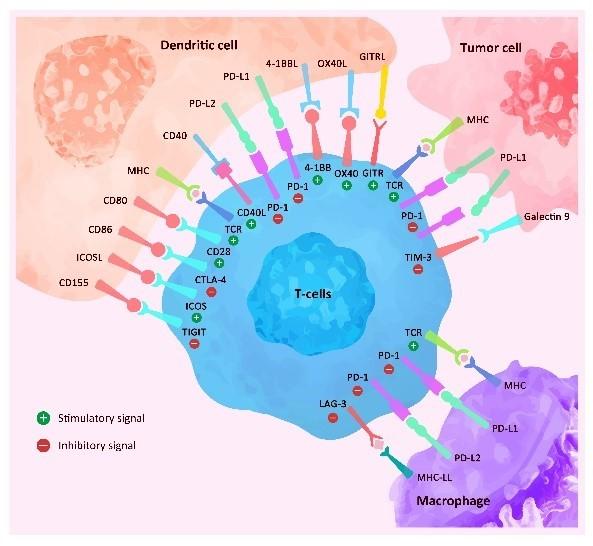
Main Applications: Immunization and antibody screening Cell-based assay Pharmacokinetic analysis
Wonder how can these immune checkpoint proteins help your drug development?
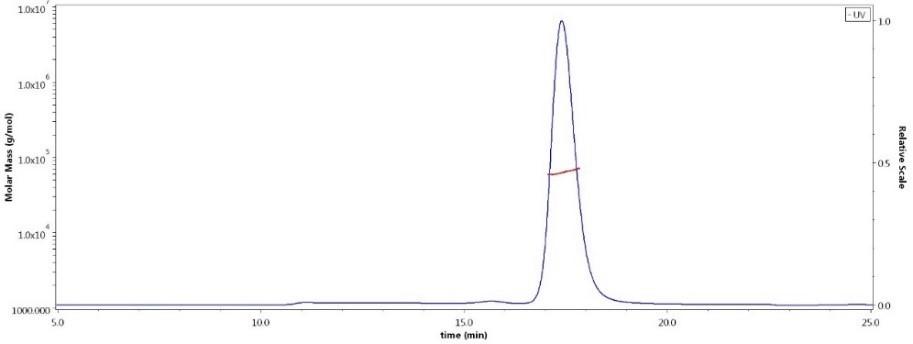
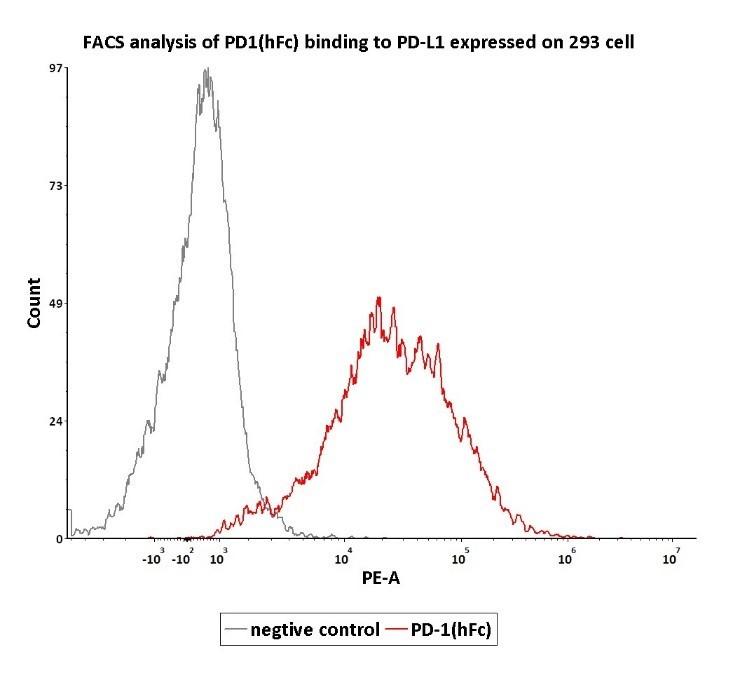
Assay Data The protocols are available for free!
|
|||||||||||||
Fount of Information は、新商品、新規取扱メーカーなどの情報をいち早く紹介するコンテンツです。情報発信のスピードを重視しているコンテンツのため、現時点で法規制や取り扱いを確認できていない商品、定価を設定できていない商品があります。ご要望やご照会を受けた商品について、法令整備や在庫の充実を図ります。