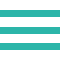Analysis of Cisplatin and its Variants in Water
Data courtesy of Prof. Ryuji Kato, Ph D., Laboratory of Cardiovascular Pharmacotherapy and Toxicology, Osaka University of Pharmaceutical Sciences
COSMOSIL πNAP uses a naphthalene-based bonded phase, and exhibits different retention behavior from C18.
In this note, πNAP is used to analyze cisplatin (CDDP) and its mono-chloro, none-chloro (hydration complex), and OH-dimer forms in water.
Standard C18 columns have difficulty separating compounds of similar hydrophobicity. Perfomance may be improved by using Cholester, a column with improved molecular shape recognition.
Experiment Summary
Cisplatin (CDDP) in water converts to its mono-chloro, hydration complex, and OH-dimer forms, and it is thought that they exist in an equilibrium state (fig. 1). Each of these forms was separated and quantified.
In water, CDDP loses Cl-, which is replaced with water. Depending on the pH, it may be replaced by a hydroxide ion. The mono-chloro form may lose another Cl-, and, in the same way, the water molecules (one or both) may be replaced with a hydroxide ion. These forms are in equilibrium.
Quantification of Cisplatin (CDDP) and its Mono-Chloro, None-Chloro, and OH-Dimer Forms
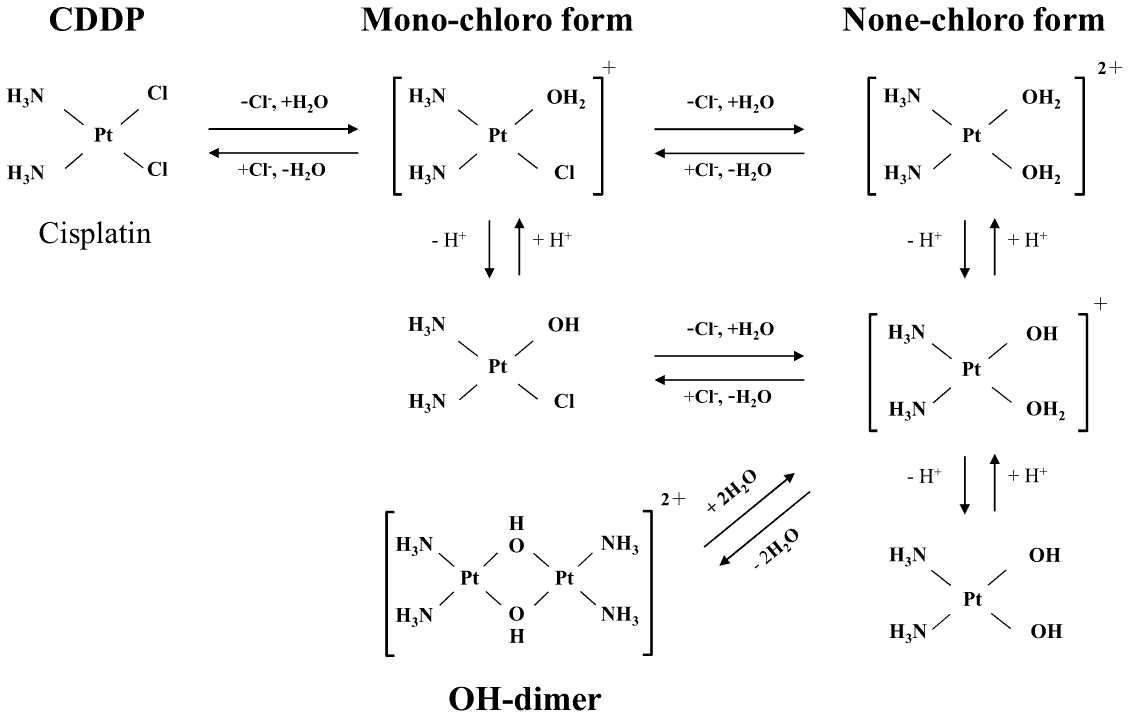
Comparing a C18 column, COSMOSIL Cholester, and πNAP under identical conditions
When measuring the OH-dimer nitrate form, the dissociated NO3- peak elutes before the OH-dimer peak. C18-MS-II could not separate the two peaks, and Cholester achieved partial separation, but could not separate to baseline. πNAP, however, was able to separate the compounds completely.
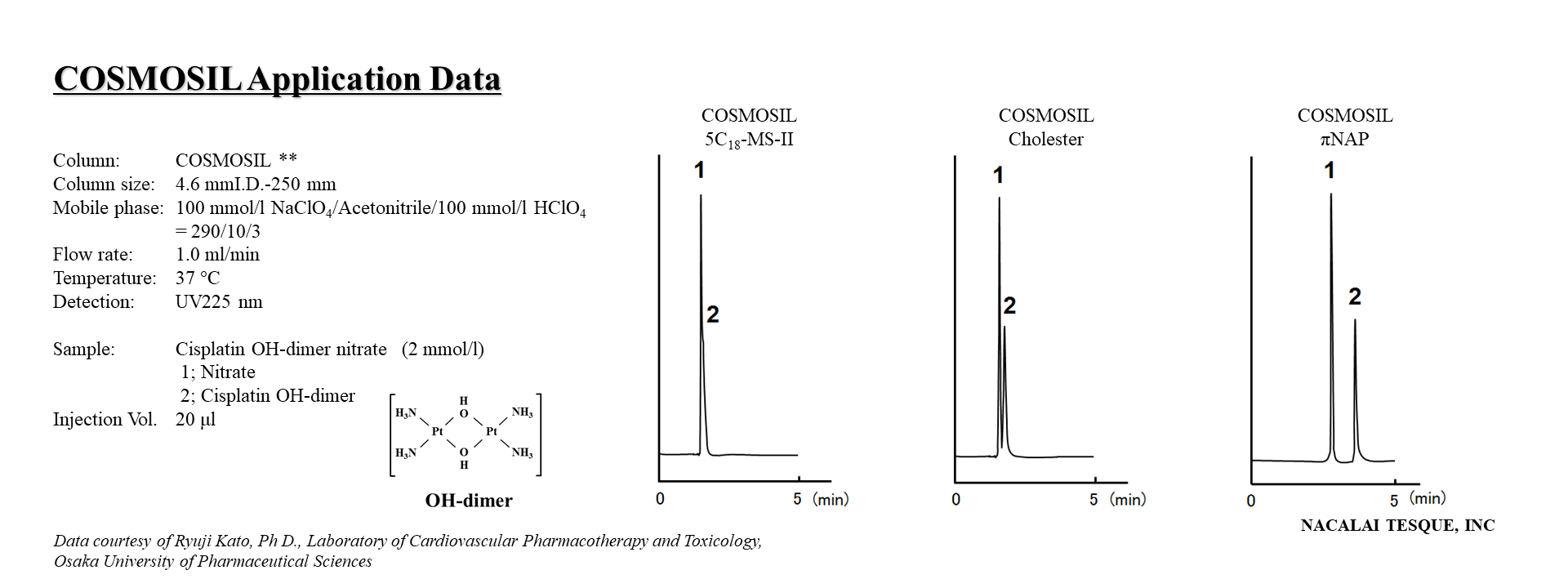
Analysis Using COSMOSIL πNAP
Below is a typical chromatogram; the peak separation was excellent. The calibration curve for cisplatin (CDDP) was linear in the range of 0.01 to 4 mM, and the CV values for within-day and between-day variation were within 5%.
Observations
By using COSMOSIL πNAP, it is possible to separate and quantify cisplatin (CDDP) and its mono-chloro, none-chloro, and OH-dimer forms using HPLC.
References
Kato R. et al. A Novel Analytical Method of Cisplatin Using the HPLC with a Naphthylethyl Group Bonded with Silica Gel (πNAP) Column. Biol Pharm Bull. 40(3), 290-296(2017). DOI